The beam size at the sample was about 1.5 × 0.4 mm ( H × V). 
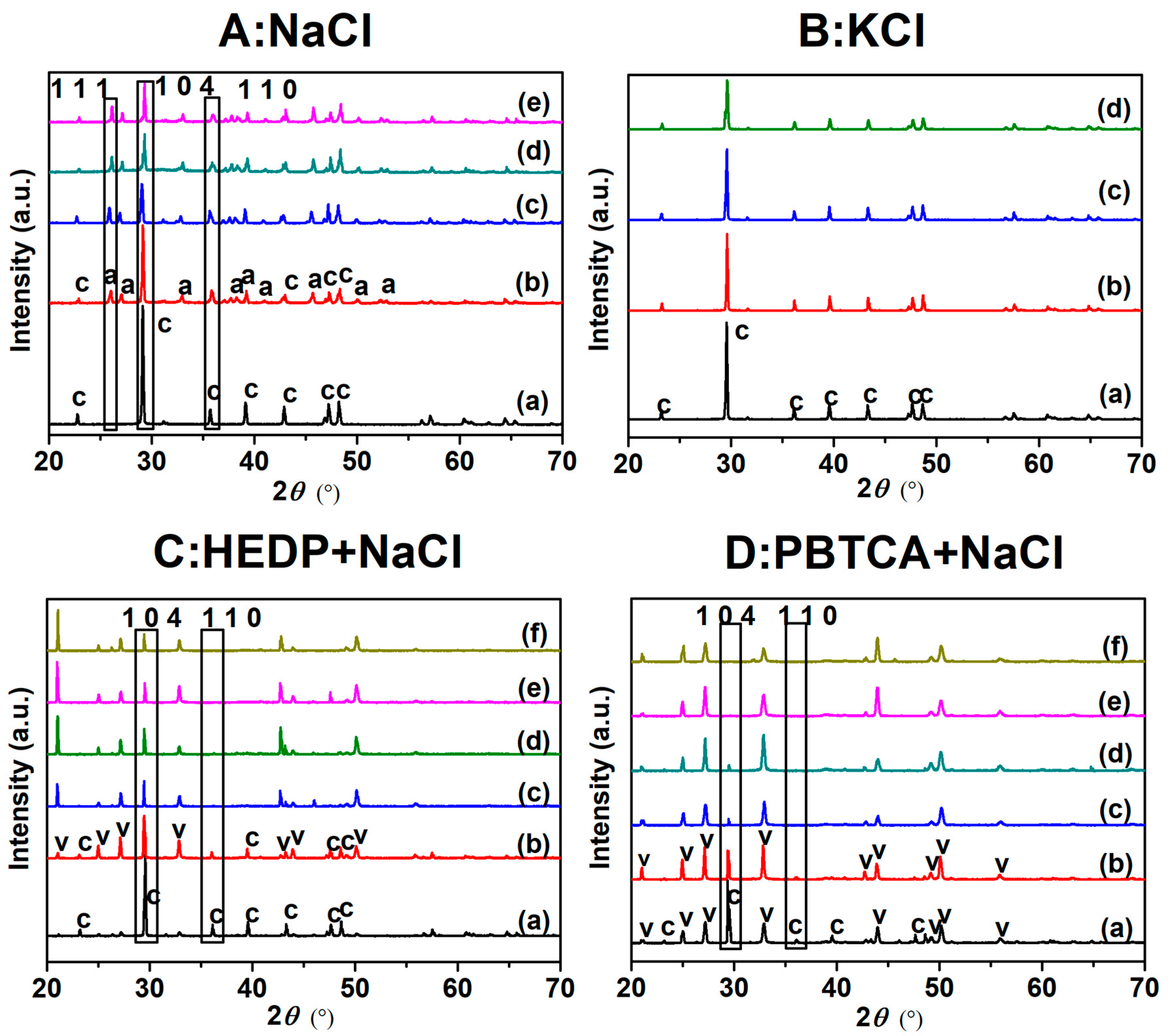
Reference standards were Nb foil as well as 0.02% NbO 2 (Aldrich) and Nb 2O 5 (Aldrich) both diluted to 0.02% in boric acid and loaded into 1 mm thick sample holders. No signs of radiation damage were detected from repeat scans, permitting multiple scans to be summed in order to improve signal-to-noise. The photon flux at the sample was around 10 10 photons s –1. The energy scale was calibrated by simultaneously measuring a Nb foil placed between the two downstream ion chambers. In the EXAFS range, k-steps of 0.035 Å –1 (up to 5 s/step) were used. Energy steps of 10 eV pre-edge and 0.35 eV across the edge (1 s/step) were used. The samples were held in a He-cooled cryostat ( T < 20 K). Spectra across the Nb K-edge ( E 0 = 18,985.6 eV (25)) were recorded in the fluorescence mode with a 100-element detector (Canberra). Samples were finely ground with a mortar and pestle and pressed into pellets. X-ray absorption spectroscopy (XAS) was performed at the wiggler XAS beamline at the Australian Synchrotron. Production of Nb-doped TiO 2 by this method may be able to supply future demand for large quantities of the material and in energy applications where a low cost of production, from readily available natural resources, would be highly desirable. Hydrolysis of Ti and Nb from acid solution, followed by calcination, leads to a well dispersed doped material, with no segregation of Nb. EXAFS for Nb demonstrates that Nb occupies a Ti site in TiO 2 with no near neighbors of Nb. 
XANES shows clear structural differences between Nb-doped anatase and rutile. Nb K-edge X-ray absorption near edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) are used to characterize the environment of 0.08 atom % Nb doped in TiO 2. Here, we investigate the structure of Nb doped in anatase and rutile produced from ilmenite digested in hydrochloric acid. When TiO 2 is precipitated from HCl solutions containing minor Nb, the Nb in solution is quantitatively deposited with the TiO 2. Niobium doping of TiO 2 creates a conductive material with many new energy applications. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
